|
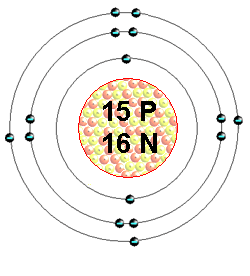
Only a couple of days updated at the moment as we will see how the week goes! Monday We will review Powers, exponents, and bases first and then will look at Base 10 and Exponent Zero Concepts. For those absent, read and perform the activities on Pages 58 and 59 to make sure we understand these concepts. Pages 61-62 questions. In Science, we will talk about the octet rule for electrons and will draw the Bohr diagrams for the first 20 elements. For this, remember: Proton number and electron number (in atoms) is equal to the atomic number. Neutron number is equal to the atomic mass (rounded to the closest whole number) - atomic number. In a Bohr diagram, we draw the nucleus first (a circle) with the number of protons and neutrons written inside. We then fill in the electrons from the inside orbital out with a maximum of 2 electrons in the first orbital, 8 in the second, 8 in the third, and 2 in the fourth (we only go up to calcium). Below is an example of phosphorus. On Tuesday, we will write a quiz for topics 1-5 in Chemistry. The notes (in addition to what has been done in class) were posted on this blog.
0 Comments
Leave a Reply. |
Mr. GordonMath/ Science 9 Archives
June 2019
Categories
All
|
 RSS Feed
RSS Feed
